General Information About Penile Cancer
Penile cancer is a disease in which malignant (cancer) cells form in the tissues of the penis.
The penis is a rod-shaped male reproductive organ that passes sperm and urine from the body. It contains two types of erectile tissue (spongy tissue with blood vessels that fill with blood to make an erection):
- Corpora cavernosa: The two columns of erectile tissue that form most of the penis.
- Corpus spongiosum: The single column of erectile tissue that forms a small portion of the penis. The corpus spongiosum surrounds the urethra (the tube through which urine and sperm pass from the body).
The erectile tissue is wrapped in connective tissue and covered with skin. The glans (head of the penis) is covered with loose skin called the foreskin. 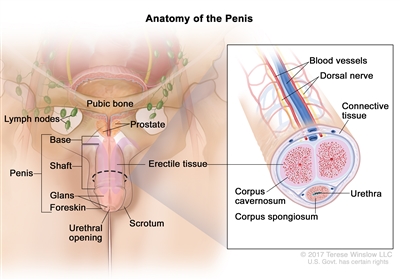
Anatomy of the penis. The parts of the penis are the base, shaft, glans, and foreskin. The tissues that make up the penis include the dorsal nerve, blood vessels, connective tissue, and erectile tissue (corpus cavernosum and corpus spongiosum). The urethra passes from the bladder to the tip of the penis.
Human papillomavirus infection may increase the risk of developing penile cancer.
Anything that increases a person's chance of getting a disease is called a risk factor. Not every person with one or more of these risk factors will develop penile cancer, and it will develop in people who don't have any known risk factors. Talk with your doctor if you think you may be at risk.
Risk factors for penile cancer include the following:
- Being uncircumcised. Circumcision may help prevent infection with the human papillomavirus (HPV). A circumcision is an operation in which the doctor removes part or all of the foreskin from the penis. Many boys are circumcised shortly after birth. Men who were not circumcised at birth may have a higher risk of developing penile cancer.
- Being age 60 or older.
- Having phimosis (a condition in which the foreskin of the penis cannot be pulled back over the glans).
- Having poor personal hygiene.
- Having many sexual partners.
- Using tobacco products.
Signs of penile cancer include sores, discharge, and bleeding.
These and other signs may be caused by penile cancer or by other conditions. Check with your doctor if you have any of the following:
- Redness, irritation, or a sore on the penis.
- A lump on the penis.
Tests that examine the penis are used to diagnose penile cancer.
In addition to asking about your personal and family health history and doing a physical exam, your doctor may perform the following tests and procedures:
- Physical exam of the penis: An exam in which the doctor checks the penis for signs of disease, such as lumps or anything else that seems unusual.
- Biopsy: The removal of cells or tissues so they can be viewed under a microscope by a pathologist to check for signs of cancer. The tissue sample is removed during one of the following procedures:
- Incisional biopsy: The removal of part of a lump or a sample of tissue that doesn't look normal.
- Excisional biopsy: The removal of an entire lump or area of tissue that doesn't look normal.
Certain factors affect prognosis (chance of recovery) and treatment options.
The prognosis and treatment options depend on the following:
- The stage of the cancer.
- The location and size of the tumor.
- Whether the cancer has just been diagnosed or has recurred (come back).
Stages of Penile Cancer
After penile cancer has been diagnosed, tests are done to find out if cancer cells have spread within the penis or to other parts of the body.
The process used to find out if cancer has spread within the penis or to other parts of the body is called staging. The information gathered from the staging process determines the stage of the disease. It is important to know the stage in order to plan treatment.
The following tests and procedures may be used in the staging process:
- CT scan (CAT scan): A procedure that makes a series of detailed pictures of areas inside the body, such as the pelvis, taken from different angles. The pictures are made by a computer linked to an x-ray machine. A dye may be injected into a vein or swallowed to help the organs or tissues show up more clearly. This procedure is also called computed tomography, computerized tomography, or computerized axial tomography.
- PET scan (positron emission tomography scan): A procedure to find malignant tumor cells in the body. A small amount of radioactive glucose (sugar) is injected into a vein. The PET scanner rotates around the body and makes a picture of where glucose is being used in the body. Malignant tumor cells show up brighter in the picture because they are more active and take up more glucose than normal cells do. When this procedure is done at the same time as a CT scan, it is called a PET/CT scan.
- MRI (magnetic resonance imaging): A procedure that uses a magnet, radio waves, and a computer to make a series of detailed pictures of areas inside the body. A substance called gadolinium is injected into a vein. The gadolinium collects around the cancer cells so they show up brighter in the picture. This procedure is also called nuclear magnetic resonance imaging (NMRI).
- Ultrasound exam: A procedure in which high-energy sound waves (ultrasound) are bounced off internal tissues or organs and make echoes. The echoes form a picture of body tissues called a sonogram.
- Chest x-ray: An x-ray of the organs and bones inside the chest. An x-ray is a type of energy beam that can go through the body and onto film, making a picture of areas inside the body.
- Biopsy: The removal of cells or tissues so they can be viewed under a microscope by a pathologist to check for signs of cancer. The tissue sample is removed during one of the following procedures:
- Sentinel lymph node biopsy: The removal of the sentinel lymph node during surgery. The sentinel lymph node is the first lymph node in a group of lymph nodes to receive lymphatic drainage from the primary tumor. It is the first lymph node the cancer is likely to spread to from the primary tumor. A radioactive substance and/or blue dye is injected near the tumor. The substance or dye flows through the lymph ducts to the lymph nodes. The first lymph node to receive the substance or dye is removed. A pathologist views the tissue under a microscope to look for cancer cells. If cancer cells are not found, it may not be necessary to remove more lymph nodes. Sometimes, a sentinel lymph node is found in more than one group of nodes.
- Lymph node dissection: A procedure to remove one or more lymph nodes in the groin during surgery. A sample of tissue is checked under a microscope for signs of cancer. This procedure is also called a lymphadenectomy.
There are three ways that cancer spreads in the body.
Cancer can spread through tissue, the lymph system, and the blood:
- Tissue. The cancer spreads from where it began by growing into nearby areas.
- Lymph system. The cancer spreads from where it began by getting into the lymph system. The cancer travels through the lymph vessels to other parts of the body.
- Blood. The cancer spreads from where it began by getting into the blood. The cancer travels through the blood vessels to other parts of the body.
Cancer may spread from where it began to other parts of the body.
When cancer spreads to another part of the body, it is called metastasis. Cancer cells break away from where they began (the primary tumor) and travel through the lymph system or blood.
- Lymph system. The cancer gets into the lymph system, travels through the lymph vessels, and forms a tumor (metastatic tumor) in another part of the body.
- Blood. The cancer gets into the blood, travels through the blood vessels, and forms a tumor (metastatic tumor) in another part of the body.
The metastatic tumor is the same type of cancer as the primary tumor. For example, if penile cancer spreads to the lung, the cancer cells in the lung are actually penile cancer cells. The disease is metastatic penile cancer, not lung cancer.
The following stages are used for penile cancer:
Stage 0
Stage 0 is divided into stages 0is and 0a.
- In stage 0is, abnormal cells are found on the surface of the skin of the penis. These abnormal cells form growths that may become cancer and spread into nearby normal tissue. Stage 0is is also called carcinoma in situ or penile intraepithelial neoplasia.
- In stage 0a, squamous cell cancer that does not spread is found on the surface of the skin of the penis or on the underneath surface of the foreskin of the penis. Stage 0a is also called noninvasive localized squamous cell carcinoma.
Stage I
In stage I, cancer has formed and spread to tissue just under the skin of the penis. Cancer has not spread to lymph vessels, blood vessels, or nerves. The cancer cells look more like normal cells under a microscope.
Stage II
Stage II is divided into stages IIA and IIB.
In stage IIA, cancer has spread:
- to tissue just under the skin of the penis. Cancer has spread to lymph vessels, blood vessels, and/or nerves; or
- to tissue just under the skin of the penis. Under a microscope, the cancer cells look very abnormal or the cells are sarcomatoid; or
- into the corpus spongiosum (spongy erectile tissue in the shaft and glans that fills with blood to make an erection).
In stage IIB, cancer has spread:
- through the layer of connective tissue that surrounds the corpus cavernosum and into the corpus cavernosum (spongy erectile tissue that runs along the shaft of the penis).
Stage III
Stage III is divided into stages IIIA and stage IIIB. Cancer is found in the penis.
- In stage IIIA, cancer has spread to 1 or 2 lymph nodes on one side of the groin.
- In stage IIIB, cancer has spread to 3 or more lymph nodes on one side of the groin or to lymph nodes on both sides of the groin.
Stage IV
In stage IV, cancer has spread:
- to tissues near the penis, such as the scrotum, prostate, or pubic bone, and may have spread to lymph nodes in the groin or pelvis; or
- to one or more lymph nodes in the pelvis, or cancer has spread through the outer covering of the lymph nodes to nearby tissue; or
- to lymph nodes outside the pelvis or to other parts of the body, such as the lung, liver, or bone.
Penile cancer can recur (come back) after it has been treated.
The cancer may come back in the penis or in other parts of the body.
Treatment Option Overview
There are different types of treatment for patients with penile cancer.
Different types of treatments are available for patients with penile cancer. Some treatments are standard (the currently used treatment), and some are being tested in clinical trials. A treatment clinical trial is a research study meant to help improve current treatments or obtain information on new treatments for patients with cancer. When clinical trials show that a new treatment is better than the standard treatment, the new treatment may become the standard treatment. Patients may want to think about taking part in a clinical trial. Some clinical trials are open only to patients who have not started treatment.
The following types of treatment are used:
Surgery
Surgery is the most common treatment for all stages of penile cancer. A doctor may remove the cancer using one of the following operations:
- Mohs microsurgery: A procedure in which the tumor is cut from the skin in thin layers. During the surgery, the edges of the tumor and each layer of tumor removed are viewed through a microscope to check for cancer cells. Layers continue to be removed until no more cancer cells are seen. This type of surgery removes as little normal tissue as possible and is often used to remove cancer on the skin. It is also called Mohs surgery.
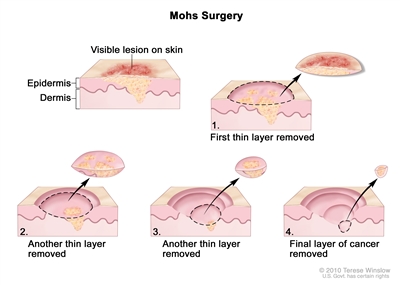
Mohs surgery. A surgical procedure to remove a visible lesion on the skin in several steps. First, a thin layer of cancerous tissue is removed. Then, a second thin layer of tissue is removed and viewed under a microscope to check for cancer cells. More layers are removed one at a time until the tissue viewed under a microscope shows no remaining cancer. This type of surgery is used to remove as little normal tissue as possible. - Laser surgery: A surgical procedure that uses a laser beam (a narrow beam of intense light) as a knife to make bloodless cuts in tissue or to remove a surface lesion such as a tumor.
- Cryosurgery: A treatment that uses an instrument to freeze and destroy abnormal tissue. This type of treatment is also called cryotherapy.
- Circumcision: Surgery to remove part or all of the foreskin of the penis.
- Wide local excision: Surgery to remove only the cancer and some normal tissue around it.
- Amputation of the penis: Surgery to remove part or all of the penis. If part of the penis is removed, it is a partial penectomy. If all of the penis is removed, it is a total penectomy.
Lymph nodes in the groin may be taken out during surgery.
After the doctor removes all the cancer that can be seen at the time of the surgery, some patients may be given chemotherapy or radiation therapy after surgery to kill any cancer cells that are left. Treatment given after the surgery, to lower the risk that the cancer will come back, is called adjuvant therapy.
Radiation therapy
Radiation therapy is a cancer treatment that uses high-energy x-rays or other types of radiation to kill cancer cells or keep them from growing. There are two types of radiation therapy:
- External radiation therapy uses a machine outside the body to send radiation toward the area of the body with cancer.
- Internal radiation therapy uses a radioactive substance sealed in needles, seeds, wires, or catheters that are placed directly into or near the cancer.
The way the radiation therapy is given depends on the type and stage of the cancer being treated. External and internal radiation therapy are used to treat penile cancer.
Chemotherapy
Chemotherapy is a cancer treatment that uses drugs to stop the growth of cancer cells, either by killing the cells or by stopping them from dividing. When chemotherapy is taken by mouth or injected into a vein or muscle, the drugs enter the bloodstream and can reach cancer cells throughout the body (systemic chemotherapy). When chemotherapy is placed directly onto the skin (topical chemotherapy), the drugs mainly affect cancer cells in those areas (regional chemotherapy). The way the chemotherapy is given depends on the type and stage of the cancer being treated.
Topical chemotherapy may be used to treat stage 0 penile cancer.
See Drugs Approved for Penile Cancer for more information.
Immunotherapy
Immunotherapy is a treatment that uses the patient's immune system to fight cancer. Substances made by the body or made in a laboratory are used to boost, direct, or restore the body's natural defenses against cancer. Topical immunotherapy with imiquimod may be used to treat stage 0 penile cancer.
New types of treatment are being tested in clinical trials.
This summary section describes treatments that are being studied in clinical trials. It may not mention every new treatment being studied. Information about clinical trials is available from the NCI website.
Radiosensitizers
Radiosensitizers are drugs that make tumor cells more sensitive to radiation therapy. Combining radiation therapy with radiosensitizers helps kill more tumor cells.
Sentinel lymph node biopsy followed by surgery
Sentinel lymph node biopsy is the removal of the sentinel lymph node during surgery. The sentinel lymph node is the first lymph node in a group of lymph nodes to receive lymphatic drainage from the primary tumor. It is the first lymph node the cancer is likely to spread to from the primary tumor. A radioactive substance and/or blue dye is injected near the tumor. The substance or dye flows through the lymph ducts to the lymph nodes. The first lymph node to receive the substance or dye is removed. A pathologist views the tissue under a microscope to look for cancer cells. If cancer cells are not found, it may not be necessary to remove more lymph nodes. Sometimes, a sentinel lymph node is found in more than one group of nodes. After the sentinel lymph node biopsy, the surgeon removes the cancer.
Treatment for penile cancer may cause side effects.
For information about side effects caused by treatment for cancer, see our Side Effects page.
Patients may want to think about taking part in a clinical trial.
For some patients, taking part in a clinical trial may be the best treatment choice. Clinical trials are part of the cancer research process. Clinical trials are done to find out if new cancer treatments are safe and effective or better than the standard treatment.
Many of today's standard treatments for cancer are based on earlier clinical trials. Patients who take part in a clinical trial may receive the standard treatment or be among the first to receive a new treatment.
Patients who take part in clinical trials also help improve the way cancer will be treated in the future. Even when clinical trials do not lead to effective new treatments, they often answer important questions and help move research forward.
Patients can enter clinical trials before, during, or after starting their cancer treatment.
Some clinical trials only include patients who have not yet received treatment. Other trials test treatments for patients whose cancer has not gotten better. There are also clinical trials that test new ways to stop cancer from recurring (coming back) or reduce the side effects of cancer treatment.
Clinical trials are taking place in many parts of the country. Information about clinical trials supported by NCI can be found on NCI's clinical trials search webpage. Clinical trials supported by other organizations can be found on the ClinicalTrials.gov website.
Follow-up tests may be needed.
Some of the tests that were done to diagnose the cancer or to find out the stage of the cancer may be repeated. Some tests will be repeated in order to see how well the treatment is working. Decisions about whether to continue, change, or stop treatment may be based on the results of these tests.
Some of the tests will continue to be done from time to time after treatment has ended. The results of these tests can show if your condition has changed or if the cancer has recurred (come back). These tests are sometimes called follow-up tests or check-ups.
Treatment of Stage 0
For information about the treatments listed below, see the Treatment Option Overview section.
Treatment of stage 0 may be one of the following:
- Mohs microsurgery.
- Topical chemotherapy.
- Topical immunotherapy with imiquimod.
- Laser surgery.
- Cryosurgery.
Use our clinical trial search to find NCI-supported cancer clinical trials that are accepting patients. You can search for trials based on the type of cancer, the age of the patient, and where the trials are being done. General information about clinical trials is also available.
Treatment of Stage I Penile Cancer
For information about the treatments listed below, see the Treatment Option Overview section.
If the cancer is only in the foreskin, wide local excision and circumcision may be the only treatment needed.
Treatment of stage I penile cancer may include the following:
- Surgery (partial or total penectomy with or without removal of lymph nodes in the groin.
- External or internal radiation therapy.
- Mohs microsurgery.
- A clinical trial of laser therapy.
Use our clinical trial search to find NCI-supported cancer clinical trials that are accepting patients. You can search for trials based on the type of cancer, the age of the patient, and where the trials are being done. General information about clinical trials is also available.
Treatment of Stage II Penile Cancer
For information about the treatments listed below, see the Treatment Option Overview section.
Treatment of stage II penile cancer may include the following:
- Surgery (partial or total penectomy, with or without removal of lymph nodes in the groin).
- External or internal radiation therapy followed by surgery.
- A clinical trial of sentinel lymph node biopsy followed by surgery.
- A clinical trial of laser surgery.
Use our clinical trial search to find NCI-supported cancer clinical trials that are accepting patients. You can search for trials based on the type of cancer, the age of the patient, and where the trials are being done. General information about clinical trials is also available.
Treatment of Stage III Penile Cancer
For information about the treatments listed below, see the Treatment Option Overview section.
Treatment of stage III penile cancer may include the following:
- Surgery (penectomy and removal of lymph nodes in the groin) with or without radiation therapy.
- Radiation therapy.
- A clinical trial of sentinel lymph node biopsy followed by surgery.
- A clinical trial of radiosensitizers.
- A clinical trial of chemotherapy before or after surgery.
- A clinical trial of new drugs or new kinds of surgery.
Use our clinical trial search to find NCI-supported cancer clinical trials that are accepting patients. You can search for trials based on the type of cancer, the age of the patient, and where the trials are being done. General information about clinical trials is also available.
Treatment of Stage IV Penile Cancer
For information about the treatments listed below, see the Treatment Option Overview section.
Treatment of stage IV penile cancer is usually palliative (to relieve symptoms and improve the quality of life). Treatment may include the following:
- Surgery (wide local excision and removal of lymph nodes in the groin).
- Radiation therapy.
- A clinical trial of chemotherapy before or after surgery.
- A clinical trial of new drugs or new kinds of surgery.
Use our clinical trial search to find NCI-supported cancer clinical trials that are accepting patients. You can search for trials based on the type of cancer, the age of the patient, and where the trials are being done. General information about clinical trials is also available.
Treatment of Recurrent Penile Cancer
For information about the treatments listed below, see the Treatment Option Overview section.
Treatment of recurrent penile cancer may include the following:
- Surgery (penectomy).
- Radiation therapy.
- A clinical trial of immunotherapy.
- A clinical trial of chemotherapy.
Use our clinical trial search to find NCI-supported cancer clinical trials that are accepting patients. You can search for trials based on the type of cancer, the age of the patient, and where the trials are being done. General information about clinical trials is also available.
To Learn More About Penile Cancer
For more information from the National Cancer Institute about penile cancer, see the following:
- Penile Cancer Home Page
- Lasers to Treat Cancer
- Cryosurgery to Treat Cancer
- Drugs Approved for Penile Cancer
- Immunotherapy to Treat Cancer
- HPV and Cancer
For general cancer information and other resources from the National Cancer Institute, see the following:
- About Cancer
- Staging
- Chemotherapy and You: Support for People With Cancer
- Radiation Therapy and You: Support for People With Cancer
- Coping with Cancer
- Questions to Ask Your Doctor about Cancer
- For Survivors and Caregivers
About This PDQ Summary
About PDQ
Physician Data Query (PDQ) is the National Cancer Institute's (NCI's) comprehensive cancer information database. The PDQ database contains summaries of the latest published information on cancer prevention, detection, genetics, treatment, supportive care, and complementary and alternative medicine. Most summaries come in two versions. The health professional versions have detailed information written in technical language. The patient versions are written in easy-to-understand, nontechnical language. Both versions have cancer information that is accurate and up to date and most versions are also available in Spanish.
PDQ is a service of the NCI. The NCI is part of the National Institutes of Health (NIH). NIH is the federal government's center of biomedical research. The PDQ summaries are based on an independent review of the medical literature. They are not policy statements of the NCI or the NIH.
Purpose of This Summary
This PDQ cancer information summary has current information about the treatment of penile cancer. It is meant to inform and help patients, families, and caregivers. It does not give formal guidelines or recommendations for making decisions about health care.
Reviewers and Updates
Editorial Boards write the PDQ cancer information summaries and keep them up to date. These Boards are made up of experts in cancer treatment and other specialties related to cancer. The summaries are reviewed regularly and changes are made when there is new information. The date on each summary ("Updated") is the date of the most recent change.
The information in this patient summary was taken from the health professional version, which is reviewed regularly and updated as needed, by the PDQ Adult Treatment Editorial Board.
Clinical Trial Information
A clinical trial is a study to answer a scientific question, such as whether one treatment is better than another. Trials are based on past studies and what has been learned in the laboratory. Each trial answers certain scientific questions in order to find new and better ways to help cancer patients. During treatment clinical trials, information is collected about the effects of a new treatment and how well it works. If a clinical trial shows that a new treatment is better than one currently being used, the new treatment may become "standard." Patients may want to think about taking part in a clinical trial. Some clinical trials are open only to patients who have not started treatment.
Clinical trials can be found online at NCI's website. For more information, call the Cancer Information Service (CIS), NCI's contact center, at 1-800-4-CANCER (1-800-422-6237).
Permission to Use This Summary
PDQ is a registered trademark. The content of PDQ documents can be used freely as text. It cannot be identified as an NCI PDQ cancer information summary unless the whole summary is shown and it is updated regularly. However, a user would be allowed to write a sentence such as "NCI's PDQ cancer information summary about breast cancer prevention states the risks in the following way: [include excerpt from the summary]."
The best way to cite this PDQ summary is:
PDQ® Adult Treatment Editorial Board. PDQ Penile Cancer Treatment. Bethesda, MD: National Cancer Institute. Updated <MM/DD/YYYY>. Available at: https://www.cancer.gov/types/penile/patient/penile-treatment-pdq. Accessed <MM/DD/YYYY>. [PMID: 26389255]
Images in this summary are used with permission of the author(s), artist, and/or publisher for use in the PDQ summaries only. If you want to use an image from a PDQ summary and you are not using the whole summary, you must get permission from the owner. It cannot be given by the National Cancer Institute. Information about using the images in this summary, along with many other images related to cancer can be found in Visuals Online. Visuals Online is a collection of more than 3,000 scientific images.
Disclaimer
The information in these summaries should not be used to make decisions about insurance reimbursement. More information on insurance coverage is available on Cancer.gov on the Managing Cancer Care page.
Contact Us
More information about contacting us or receiving help with the Cancer.gov website can be found on our Contact Us for Help page. Questions can also be submitted to Cancer.gov through the website's E-mail Us.
Last Revised: 2023-05-12
If you want to know more about cancer and how it is treated, or if you wish to know about clinical trials for your type of cancer, you can call the NCI's Cancer Information Service at 1-800-422-6237, toll free. A trained information specialist can talk with you and answer your questions.